UK TEL: +44 (0) 1234 780020 | USA TEL: +1 888 381 6870
World class contract research services
Your point-of-care tests powered by innovative science.
Global Access Diagnostics (GADx) offers a complete range of services to researchers, developers and manufacturers of rapid point-of-care assays and devices.
Our team of more than 40 scientists has extensive experience in dealing with simplex or multiplex tests, device housing and product design, both for unique test systems and for our innovative enzyme activity detection (ELTABA) platform.
We can help with everything from identifying the right proteins and antibodies, to designing the most effective device and scaling up low- to mid-volume manufacture.
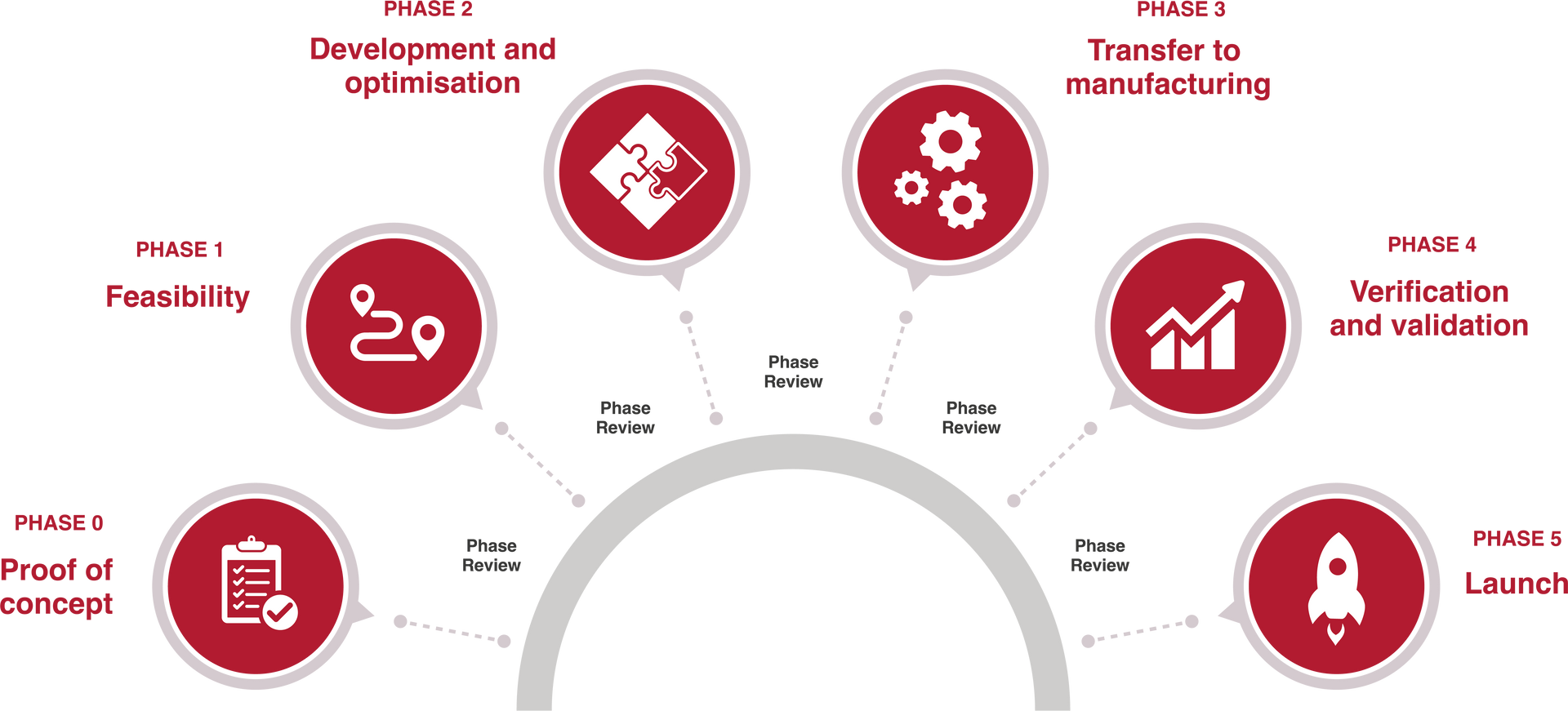
All of our work is carefully stage-gated, from proof of concept to launch. We review progress together and ensure we're making the right decisions to develop your assay or product.
Assay Development

Global Access Diagnostics offers a complete range of services to researchers, developers and manufacturers of rapid point-of-care assays and devices.
Our team of more than 40 scientists has extensive experience in dealing with simplex or multiplex tests, device housing and product design, both for unique test systems and for our innovative enzyme activity detection (ELTABA) platform.
We can help with everything from identifying the right proteins and antibodies to designing the most effective device ready for manufacturing transfer.
All of our work is carefully stage-gated, from proof of concept to launch. We review progress together and ensure we're making the right decisions to develop your assay or product.
REAGENT DEVELOPMENT
GADx has produced hundreds of rare biomolecules for clients and for our own in-house development. Our expertise is in the production of highly specific and well characterized reagents including those used in immunoassays where performance is directly related to reagent sensitivity, specificity and purity. We have deep experience in developing or sourcing high affinity antibodies and controlling methods for production of recombinant Fab binding molecules that are directed toward your analyte of interest. In addition, we’ve developed methods for increasing the immunogenicity of your antigen with the inclusion of virus like particles (VLP) in the immunization strategy.

We can also provide epitope mapping of the antigen to confirm the specificity of the response. GADx offers peptide and antigen synthesis, nanoparticle / enzyme conjugation and enzyme substrate optimization. GADx also provides additional reagent development services related to:
- Epitope design (rational)
- Recombinant antibody production
- Traditional Polyclonal and monoclonal antibody development
- Recombinant protein production (E. coli and yeast)
- Microbiology: prokaryote and lower eukaryote (class I and II organisms)
REAGENT ENRICHMENT / PROCESS DEVELOPMENT

Affinity purification is an ideal way to increase reagent specificity. GADx can provide detailed purification strategies or deliver highly purified material based on your target antigen. We offer additional process development services relating to enrichment of recombinant proteins and monoclonal antibodies. Our experience in immunoassay design has allowed us to optimize and develop procedures for biomolecule conjugation, buffered surfactants and scale-up for manufacturing.
REAGENT ANALYSIS
Impure reagents can lead to downstream design flaws and finished product instability, so it is crucial we ensure reagents are free of contaminants. GADx has invested heavily in both staff and equipment capability to fully assess reagent purity. To do this, we utilize chromatography tools (ex. FPLC, HPLC), electrophoresis tools (ex. SDS-PAGE/ Western blot) and spectrometry tools (ex. UV/VIS, fluorimetry, dynamic light scattering, and mass spectrometry).
The characterization of biomolecule activity is paramount to its utility and allows for appropriate downstream inclusion. Our capability uses information gathered from peptide epitope mapping,

bioactivity testing, flow cytometry / imaging microscopy and label free kinetic analysis (which is superior to SPR results) to optimize the reagent’s ability to exceptionally function in the finished product.
Our development packages (immunoassay, antibody, recombinant protein) include a range of analytical testing methods to ensure quality and activity of critical reagents specifically relating to identity, purity, and stability. We are also able to offer reagent analysis as a standalone service to provide characterization for already developed materials.
DEVICE FORMAT DEVELOPMENT

Immunoassays come in many shapes, sizes, and forms. GADx has been involved in them all, from microwell EIA testing, to dipstick lateral flow tests, to fully integrated over the counter disposable systems. Our experience can help determine proper detection strategy (enzymatic, electrochemical, visual nanoparticle) as well as test through-put. We also have the ability for rapid prototyping (3D printing) to create custom housing that can integrate sample collection and the processing steps.
GADx also offers services related to laser etching and freeze dry lyophilization. In addition, our patented enzyme activity detection technology (ELTABA) is available for use.
EVALUATION AND VALIDATION SERVICES
GADx's core competency is in delivering immunoassays that meet a market need. This involves verifying clinical utility with confirmed samples, characterization and elimination of sample matrix effects, and product shelf-life stability. These important activities are included with any of our contract development offerings and can be utilized separately, for your specific needs. In addition, we offer consulting services relating to human factor usability, up-stream marketing and voice of the customer. We are ISO 13485:2016 and MDSAP approved and can provide IQ/OQ/PQs, design history files, batch records and other required regulatory documentation. We have strong links to the clinical community and have undertaken numerous trials, so we can facilitate small scale clinical trials as well as training sets and user simulation sessions.

CONNECTED DIAGNOSTICS

GADx is committed to enabling tests to be digitally connected to support more accurate diagnoses and better use of data to support surveillance and other public health needs. We can work with you ensure the tests we develop together are readable by a variety of readers, and can help you select the right partners to develop companion apps, middleware and other data management services.
WHO BENEFITS FROM GADx CONTRACT RESEARCH SERVICES?
Diagnostic companies
You are looking for innovation and scientific excellence to help advance your R&D agenda and ensure your devices are the best on the market.
Pharma and Life Sciences Companies
You are looking for a companion diagnostic to better serve your target population, enabling them to understand their health status and empowering them to take action on it through a service or treatment you provide.
Global health R&D funders
You are looking for a company that prioritizes impact to address a pressing global health need, safe in the knowledge that our incentives are aligned to continue to provide products with access and impact prioritized above profit.
CONCEPT TO CUSTOMER MADE EASY
Exploration
Let’s find out if we’re the best fit. We discuss your needs and explore the best possibilities to meet them. This no-obligation consulting phase will lead to a Scope of Work defining technical, regulatory and market requirements, resulting in a quote.
Prototyping and validation
Upon your approval we continue to the initial production and verification phase with carefully structured stage gating, excellent project management and ongoing quality assurance.
Production
When all requirements are met we transfer to manufacturing and will meet your objectives in flexible volume, time, quality and cost.
Distribution
We ship to you directly or can help you distribute your product through our established international distribution network.
GLOBAL HEALTH MADE ACCESSIBLE
TELL US WHAT YOU NEED
Get in touch to find out how we can help meet your rapid, point-of-care diagnostics needs.
Tell Us What You Need - Contract Research
We will get back to you as soon as possible.
Please try again later.
Mologic Ltd trading as Global Access Diagnostics. Company registered in England and Wales .Company Registration No. 4784437 | All Rights Reserved



